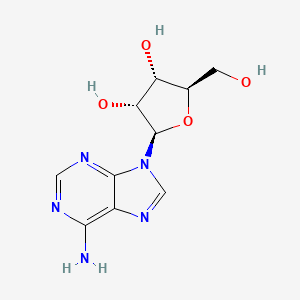
Chemical Structure
Learn about Adenosine — its natural origin index (ISO 16128), feedstock sources, synthesis process, and renewability classification.
View full data on PubChem ↗Quick Facts
- INCI NameAdenosine
- IUPAC Name(2R,3R,4S,5R)-2-(6-aminopurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol
- Molecular FormulaC10H13N5O4
- PubChem CID60961 ↗
- FunctionsSkin Conditioning AgentAntioxidant
- Primary FeedstockThe primary commercial feedstock for adenosine synthesis is derived from petrochemical sources, typically involving the production of purine bases and ribose moieties from simpler organic molecules. While alternative biological routes are explored, the dominant industrial pathway relies on synthetic chemistry originating from fossil fuels.
- NOI Score0.0 / 1.0
- Tier100% Petroleum
Description
Learn about Adenosine — its natural origin index (ISO 16128), feedstock sources, synthesis process, and renewability classification.
References
- 1ISO 16128-2:2017 — Guidelines on technical definitions and criteria for natural and organic cosmetic ingredients and products — Part 2. ISO, Geneva.
- 2Adenosine. National Center for Biotechnology Information. PubChem CID 60961. pubchem.ncbi.nlm.nih.gov ↗